The Power of Potassium

Knowing The Initial Potassium Concentration Can Transform Your Winemaking Techniques
In the pursuit of exceptional wine, understanding the composition of your grape must is essential. One element that has garnered increasing attention—especially among East Coast winemakers—is potassium (K+). While West Coast producers have long recognized its influence on acidity and stability, recent research from the Virginia Winemakers Research Exchange (WRE) has shed new light on potassium’s complex role in shaping a wine’s balance, mouthfeel, and aging potential.
In this article, we explore how potassium interacts with tartaric acid, affects pH, and what you can do—practically and scientifically—to make informed decisions throughout your winemaking process.
Why Test for Potassium?

Potassium (K⁺) is the most abundant cation found in grapes and plays a crucial role in wine chemistry, particularly in:
- pH modulation
- Tartaric acid stability
- Bitterness perception
- Tartrate precipitation
Excess potassium can raise the pH of your wine, reduce titratable acidity (TA), and thus increase the risk of microbial instability. Testing grape juice for potassium provides early insight into how the must will behave during fermentation, stabilization, and aging.
Key benefits of potassium testing:
- Anticipate pH shifts before fermentation.
- Predict tartrate stability and cold stabilization needs.
- Tailor acid additions to maintain balance and microbial safety.
How Potassium Impacts Wine Chemistry
When potassium is present in high concentrations, it binds with tartaric acid to form potassium bitartrate (KHT). This compound is less soluble at lower temperatures and precipitates out—especially during cold stabilization—resulting in a loss of acidity and an increase in pH.
Fun Fact
Potassium bitartrate is better known to the world as cream of tartar! Commercial cream of tartar is made by processing this winemaking byproduct on a mass scale.

Common Effects of High Potassium in Must:
| Effect | Winemaking Implication |
|---|---|
| Increased pH | Diminished freshness, greater microbial risk |
| Reduced TA | Flatter mouthfeel and less structural integrity |
| Greater KHT precipitation | More aggressive cold stabilization needed |
| Delayed malolactic fermentation | Altered sensory development |
Understanding the Potassium-Tartaric Acid Equilibrium
At the heart of this issue is the acid-base equilibrium between tartaric acid and potassium. Tartaric acid exists in pH-dependent equilibrium with its dissociated forms (HT⁻ and T²⁻). Potassium interacts primarily with the bitartrate anion (HT⁻) to form KHT, which is poorly soluble in alcoholic solutions (aka wine) and precipitates easily, especially during cooling.
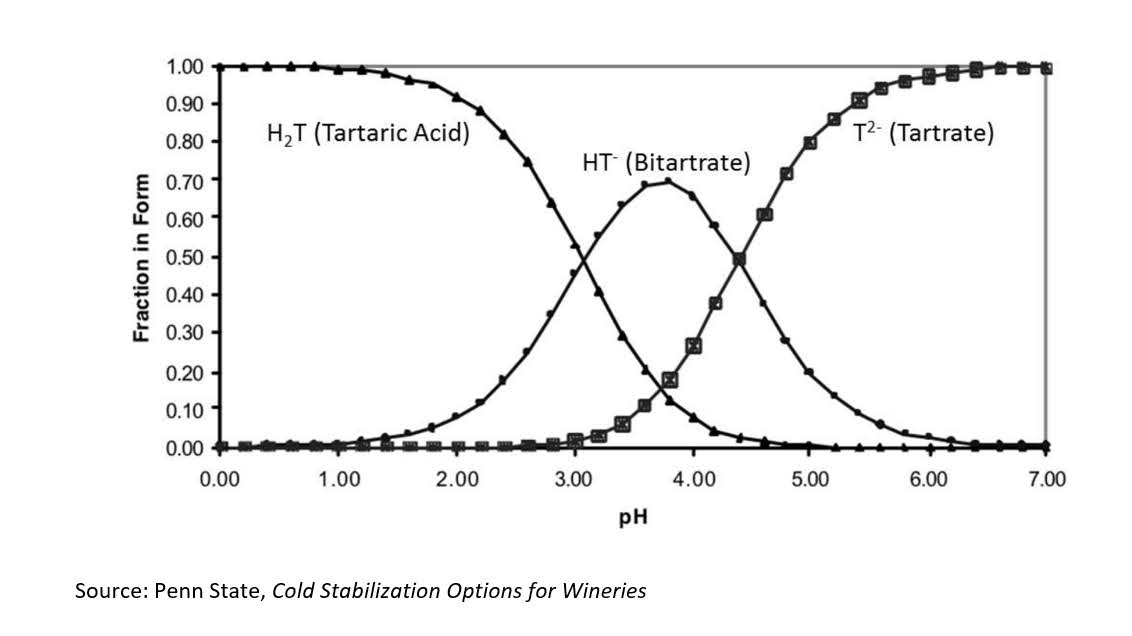
This precipitation causes a decline in titratable acidity (less tartaric acid) but doesn’t remove protons (H+), so it can actually increase pH despite an apparent loss of acid.
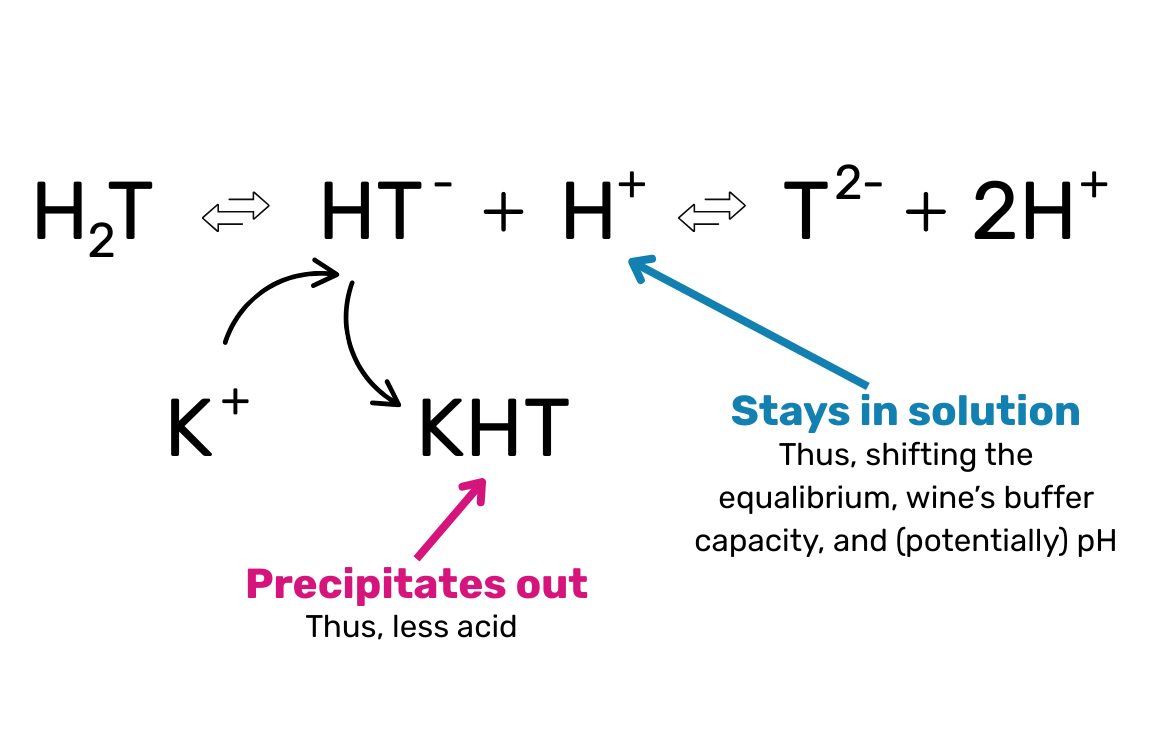
Why this matters:
As the pH increases beyond 3.65–3.80, the wine becomes significantly less stable both chemically and microbially. This critical range is where winemakers must be most vigilant.
Why pH 3.8 Is a Tipping Point
Around pH 3.8, a shift occurs in the chemical equilibrium that dramatically increases the precipitation of KHT. This level is often seen as a threshold because:
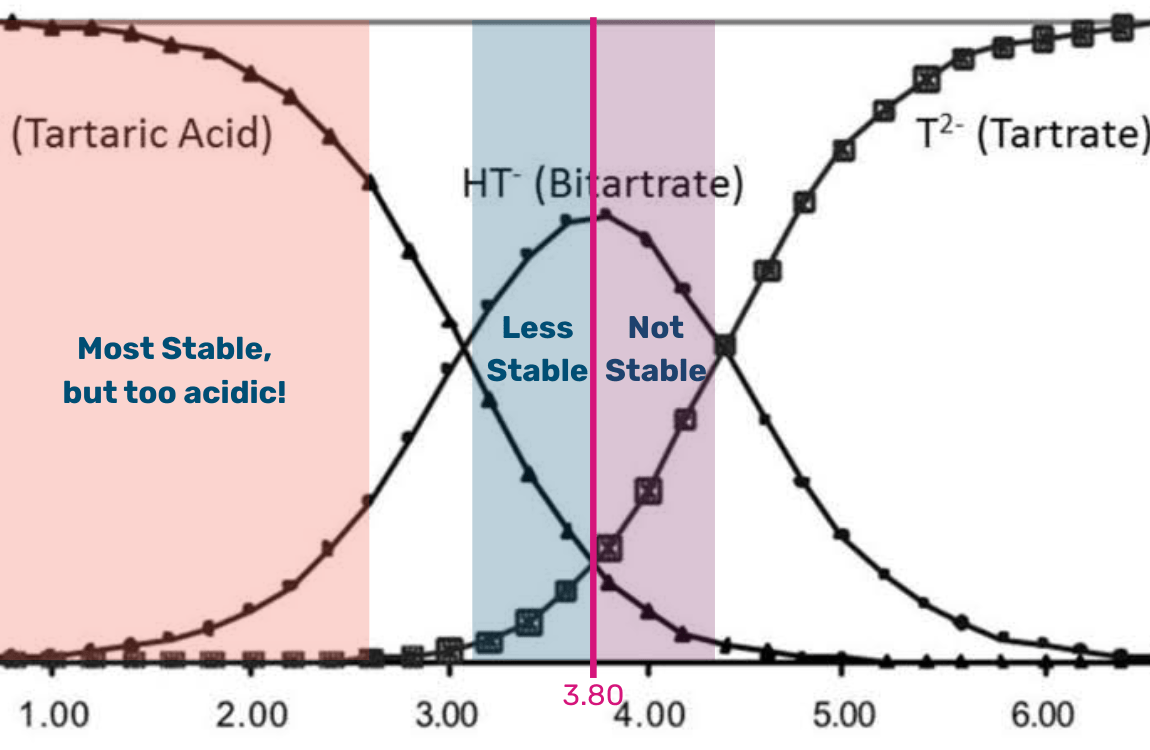
- Above pH 3.8: most of the tartaric acid is deprotonated and available to bind with potassium.
- Below pH 3.6: precipitation slows and tartrate stability improves.
Takeaway: If your juice or wine is nearing or above this threshold, cold stabilization, acidification, or blending may be necessary to avoid downstream quality issues.
Practical Applications for Winemakers
1. Pre-Fermentation Grape Juice Testing
Before fermentation begins:
- Test for potassium, pH, TA, and malic acid.
- Segment vineyard lots to understand variability.
2. Adjusting Acid Levels with Knowledge
If potassium is high:
- Add tartaric acid pre-fermentation, when it’s most stable and effective.
- Avoid post-fermentation additions unless needed for stability, as pH effects may be limited.
- Consider using cold stabilization to manage excess KHT before bottling.
3. Monitor Throughout Aging
Track potassium and pH changes during:
- Malolactic fermentation (MLF) — can further raise pH.
- Cold stabilization trials — ensure tartrate stability.
- Pre-bottling assessments — avoid haze, instability, and bottle sediment.
Potassium Levels in Common Varieties
Potassium content varies widely by variety, terroir, and vineyard management. Here are average levels found in U.S. musts:
| Variety | Potassium (mg/L) |
|---|---|
| Chardonnay | 900-1,200 |
| Cabernet Sauvignon | 1,000-1,300 |
| Merlot | 950-1,200 |
| Riesling | 800-1,000 |
| Shiraz / Syrah | 1,200-1,500 |
| Pinot Noir | 850-1,100 |
High potassium levels (>1,200 mg/L) are more common in warmer regions or low-acid vineyards and should be addressed proactively.
Virginia-Specific Data: WRE Findings
The Virginia Winemakers Research Exchange has contributed important regional data:
- Potassium levels in some Virginia vineyards are among the highest in the U.S.
- Soil type, especially high-clay or calcareous soils, correlates with elevated potassium uptake.
- Acidification strategies are more frequently needed due to climatic and varietal factors.
Collaborative studies have shown that early juice testing and targeted acid additions significantly improve wine quality, color retention, and shelf stability.

Conclusion: Test Early, Adjust Intelligently
Whether you’re growing vinifera in Virginia or hybrid grapes in the Finger Lakes, potassium is a key player in your wine’s final expression. Armed with data and a clear understanding of how K⁺ interacts with acids, you can proactively shape fermentation outcomes, improve stability, and produce more age-worthy wines.
Winemaker Action Plan:
- 🍇 Test must pre-fermentation for K⁺, pH, TA, and Malic Acid.
- 📉 Watch for pHs greater than 3.6–3.8.
- 🧪 Use tartaric acid early when needed to avoid overcorrection post-fermentation.
- ❄️ Cold stabilize when required, especially for high K⁺ wines.
- 📊 Keep records vintage to vintage to refine your practices.
Further Reading & Resources
- Penn State Extension: Cold Stabilization Options for Wineries
- Virginia Winemakers Research Exchange
- UC Davis Viticulture & Enology Department
Let’s raise a glass to better chemistry, cleaner balance, and wine that sings with structure and stability. 🍷
